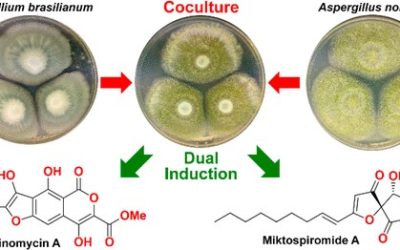
Shengping Zhang, Luis M. De Leon Rodriguez, Ernest Lacey, Andrew M. Piggott, Ivanhoe K. H. Leung, Margaret A. Brimble
Europ. JOC, 2017, 149.
Publication Date: November 7, 2016
https://doi.org/10.1002/ejoc.201601016
Abstract:
The synthesis of a series of naturally occurring cyclotetrapeptides and an analogue with 1-propanephosphonic acid anhydride (T3P) is reported. An unexpected finding is the formation of unstable cyclic peptide conformers in addition to the more stable trans,cis,trans,cis (tctc) cyclopeptides.
The cyclization of the linear tetrapeptides sequence H-NMePhe-Xaa1–NMePhe-Xaa2-OH (for which Xaa1 = Xaa2 = Ile, Val, or Leu; Xaa1 = Val; Xaa2 = Ile or Leu) by using 1-propanephosphonic acid anhydride was explored. An unanticipated finding is the cyclization towards cyclotetrapeptide conformers (kinetic products) that were highly prone to hydrolysis and that slowly interconvert into the more stable trans,cis,trans,cis (tctc) conformers (thermodynamic products).