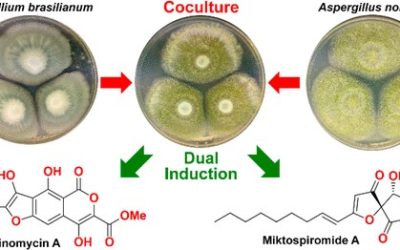
Monika Kohnke, Steven Schmitt, Nicholas Ariotti, Andrew M. Piggott, Robert G. Parton, Ernest Lacey,
Robert J. Capon, Kirill Alexandrov, and Daniel Abankwa
Chemistry and Biology 2012, 19, 866-74.
Publication Date: May 30, 2012
https://doi.org/10.1016/j.chembiol.2012.05.019
Abstract:
Protein prenylation is required for membrane anchorage of small GTPases. Correct membrane targeting is essential for their biological activity. Signal output of the prenylated proto-oncogene Ras in addition critically depends on its organization into nanoscale proteolipid assemblies of the plasma membrane, so called nanoclusters. While protein prenylation is an established drug target, only a handful of nanoclustering inhibitors are known, partially due to the lack of appropriate assays to screen for such compounds. Here, we describe three cell-based high-throughput screening amenable Förster resonance energy transfer NANOclustering and Prenylation Sensors (NANOPS) that are specific for Ras, Rho, and Rab proteins. Rab-NANOPS provides the first evidence for nanoclustering of Rab proteins. Using NANOPS in a cell-based chemical screen, we now identify macrotetrolides, known ionophoric antibiotics, as submicromolar disruptors of Ras nanoclustering and MAPK signaling.